Figure 6: Svante Arrhenius deciphered the ionization process when a salt is dissolved in a solvent. This discovery made it clear that the ionization process isn’t mediated only by the passage of electric current. He was honored with the Nobel Prize in Chemistry in 1903. 
The discovery of this salt dissociation phenomenon upon its dissolution was explained by Svante Arrhenius in his 1884 dissertation. When a solid crystalline salt is dissolved in an aqueous medium, the charged entities are dissociated as separate particles (Faraday’s ions). Image Credit: SciHi Blog.ĭiscovery of the Salt Dissociation Process upon Dissolution Figure 5: William Whewell coined several terms including anode, cathode, anion, and cation. The terms anode, cathode, cation, and anion were coined by William Whewell. Coining of The Terms Anode, Cathode, Anion, and Cation Figure 4: Michael Faraday coined the term ion. As observed, electrode (metal) dissolution at one end and the new metal formation (electrode increasing) at another end certainly had some mediating factors. An important observation of Faraday was the ability of some “medium species” to travel across the aqueous solution from one electrode to another. It was only known as the species traveling from one electrode to another electrode both electrodes being present in an aqueous medium. At that time, the ionic concept and the nature of the chemical species as it is known today weren’t clear. The term ion was coined in 1834 by English physicist cum chemist Michael Faraday. Let’s discuss a brief historical aspect of ions, the coining of various terms, and scientific discoveries. These are examples of some ionic compounds. Figure 3: Ionic compounds are formed when anions and cations come together and establish an ionic bond. What is an ionic compound? Since opposite charges attract each other, anions and cations being negatively and positively charged respectively, readily come together to form ionic compounds. Now you can tell what is a monatomic ion and what are polyatomic ions… Image Credit: Expii. Consisting of two or more atoms: POLYATOMIC ION / MOLECULAR IONįigure 2: Difference between monoatomic and polyatomic ion formula and monoatomic and polyatomic ions examples.Consisting of a single atom: MONOATOMIC ION (an ion made of a single atom).Image Credit: The Bumbling Biochemist.īased on the number of atoms involved, ions are of two types: Negatively charged ions and negatively charged particles are called anions (negative ions).įigure 1: A representation of ion concept and difference between anion and cation. Negatively charged atoms & Fewer protons 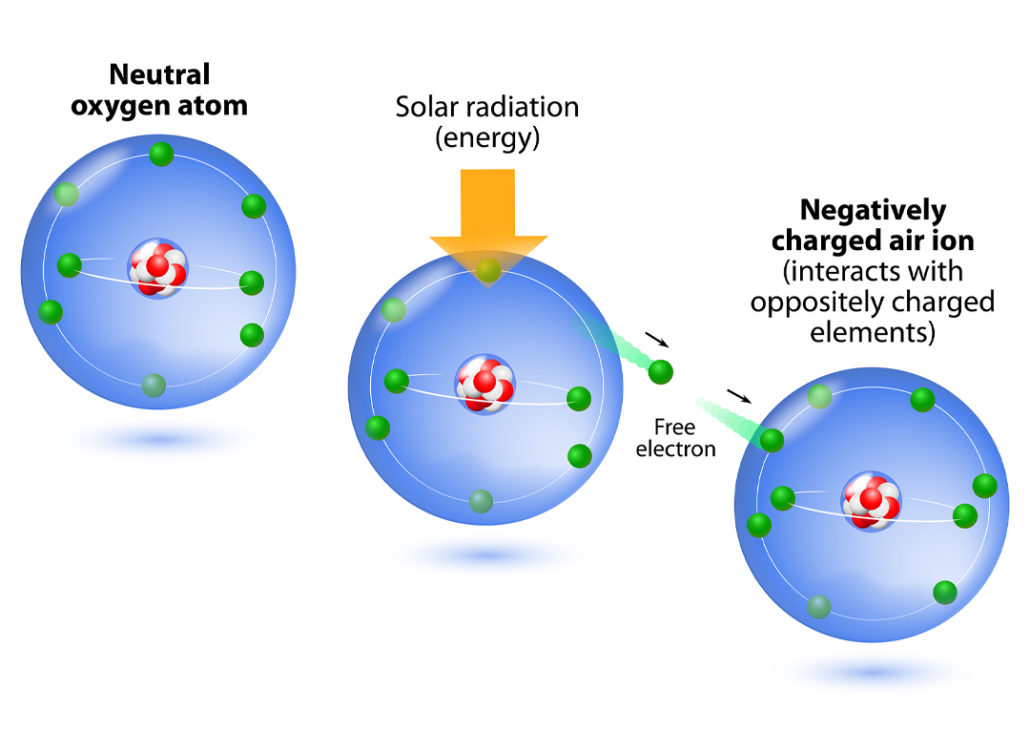
Sodium ions and calcium ions are examples of cations. Positively charged ions and positively charged particles are called cations. Positively charged atoms & More protons > Fewer electrons: CATION (formed by loss of electrons in an atom).Based on the type of charge and the ratio of protons and electrons, ions are of two types:
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
