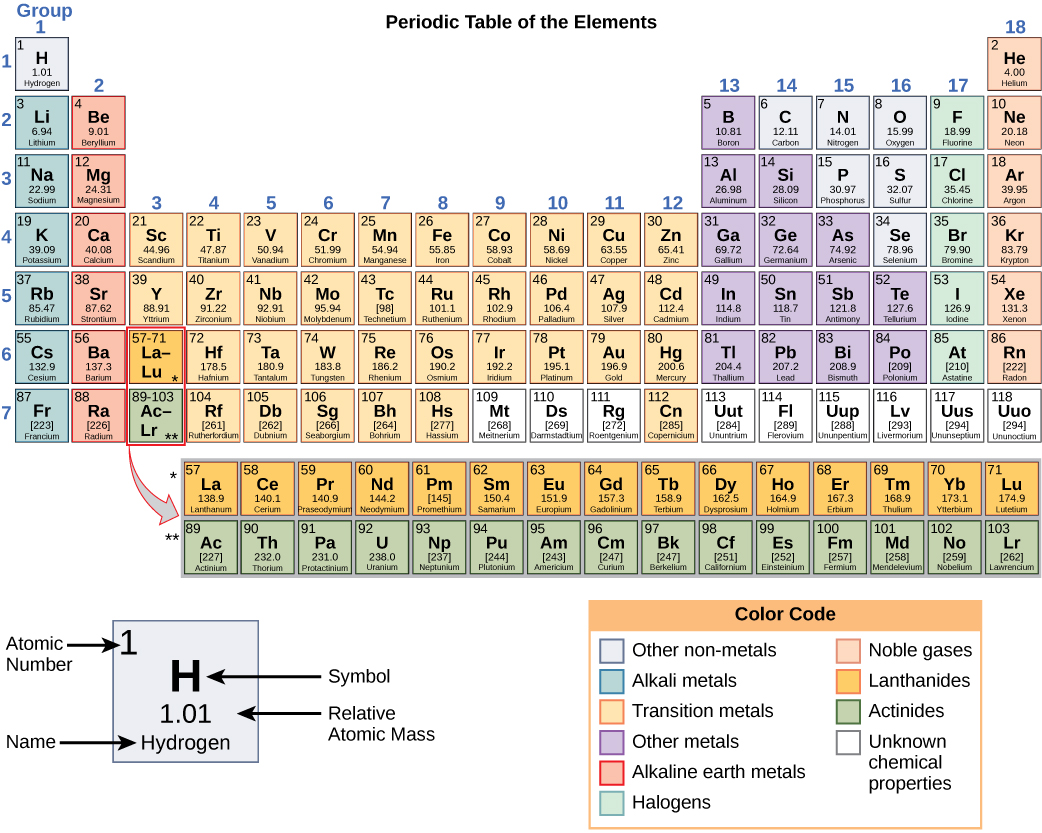
To make iodine line up with chlorine and bromine in his table, Mendeleev swapped the positions of iodine and tellurium. However, iodine has similar chemical properties to chlorine and bromine. So iodine should be placed before tellurium in Mendeleev's tables. Iodine has a lower relative atomic mass than tellurium. Its properties were found to be similar to the predicted ones and confirmed Mendeleev's periodic table. The element germanium was discovered later. For example, Mendeleev predicted the existence of 'eka-silicon', which would fit into a gap next to silicon. By looking at the chemical properties and physical properties of the elements next to a gap, he could also predict the properties of these undiscovered elements. Mendeleev left gaps in his table to place elements not known at the time. Mendeleev's periodic table Predictions using gaps he switched the order of a few elements to keep the groups consistent.he left gaps for yet to be discovered elements.To make his classification work Mendeleev made a few changes to his order: He then arranged the elements by putting those with similar properties below each other into groups. atomic number 12 atomic weight 24. Its compounds are widely used in construction and medicine, and magnesium is one of the elements essential to all cellular life. When he did this he noted that the chemical properties of the elements and their compounds showed a periodic trend. magnesium (Mg), chemical element, one of the alkaline-earth metals of Group 2 (IIa) of the periodic table, and the lightest structural metal. Mendeleev arranged the elements in order of increasing relative atomic mass. For more detailed information about the origins of element names, see List of chemical element name etymologies. Mendeleev published his first periodic table of the elements in 1869. Like the periodic table, the list below organizes the elements by the number of protons in their atoms it can also be organized by other properties, such as atomic weight, density, and electronegativity. Like many scientists working at the end of the 19th-century the Russian chemist Dmitri Mendeleev (1834-1907) was looking for ways to organise the known elements. Mendeleev's periodic table Dmitri Mendeleev
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
