
It features a functional section on how the body works, system by system. The Complete Human Body begins with a region-by-region anatomical atlas, containing extra detail on the hands, feet, and major joints. Diseases and disorders section includes easy-to-follow summaries of over 200 health conditions. Feature panels and spreads throughout explore fascinating aspects of the human body. A functional section then shows how the body works, system by system. Dive into the pages of this brilliant book on the human body, to discover: - A region-by-region anatomy atlas allows the reader to explore the body up close and almost life-size from head to toe. Guided by Professor Alice Roberts, renowned academic, author, and broadcaster, and groundbreaking 3-D computer-generated illustrations, you will understand the human body as never before, explore human anatomy in incredible detail and clarity, and discover how the body works, how it changes from infancy to old age, and what can go wrong and why. Although used by students and healthcare professionals, it is accessible enough for anyone to dip into and enjoy. The most detailed popular reference book on human anatomy available, this beautiful exploration of the human body is now in its third edition, revised with the latest medical knowledge. The definitive guide to the development, form, function, and disorders of the human body. To learn more about how and for what purposes Amazon uses personal information (such as Amazon Store order history), please visit our Privacy Notice. 
You can change your choices at any time by visiting Cookie Preferences, as described in the Cookie Notice. Click ‘Customise Cookies’ to decline these cookies, make more detailed choices, or learn more. 
Third parties use cookies for their purposes of displaying and measuring personalised ads, generating audience insights, and developing and improving products. This includes using first- and third-party cookies, which store or access standard device information such as a unique identifier. If you agree, we’ll also use cookies to complement your shopping experience across the Amazon stores as described in our Cookie Notice. We also use these cookies to understand how customers use our services (for example, by measuring site visits) so we can make improvements. We use cookies and similar tools that are necessary to enable you to make purchases, to enhance your shopping experiences and to provide our services, as detailed in our Cookie Notice.
0 Comments
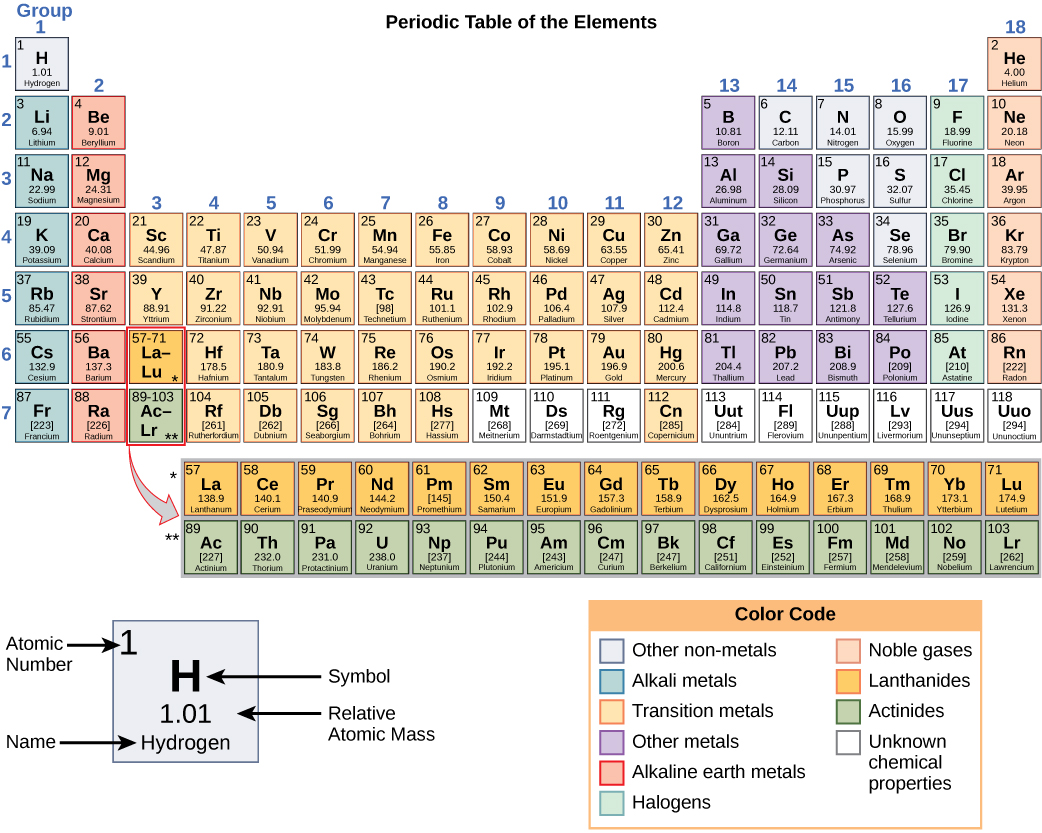
To make iodine line up with chlorine and bromine in his table, Mendeleev swapped the positions of iodine and tellurium. However, iodine has similar chemical properties to chlorine and bromine. So iodine should be placed before tellurium in Mendeleev's tables. Iodine has a lower relative atomic mass than tellurium. Its properties were found to be similar to the predicted ones and confirmed Mendeleev's periodic table. The element germanium was discovered later. For example, Mendeleev predicted the existence of 'eka-silicon', which would fit into a gap next to silicon. By looking at the chemical properties and physical properties of the elements next to a gap, he could also predict the properties of these undiscovered elements. Mendeleev left gaps in his table to place elements not known at the time. Mendeleev's periodic table Predictions using gaps he switched the order of a few elements to keep the groups consistent.he left gaps for yet to be discovered elements.To make his classification work Mendeleev made a few changes to his order: He then arranged the elements by putting those with similar properties below each other into groups. atomic number 12 atomic weight 24. Its compounds are widely used in construction and medicine, and magnesium is one of the elements essential to all cellular life. When he did this he noted that the chemical properties of the elements and their compounds showed a periodic trend. magnesium (Mg), chemical element, one of the alkaline-earth metals of Group 2 (IIa) of the periodic table, and the lightest structural metal. Mendeleev arranged the elements in order of increasing relative atomic mass. For more detailed information about the origins of element names, see List of chemical element name etymologies. Mendeleev published his first periodic table of the elements in 1869. Like the periodic table, the list below organizes the elements by the number of protons in their atoms it can also be organized by other properties, such as atomic weight, density, and electronegativity. Like many scientists working at the end of the 19th-century the Russian chemist Dmitri Mendeleev (1834-1907) was looking for ways to organise the known elements. Mendeleev's periodic table Dmitri Mendeleev |
 RSS Feed
RSS Feed
